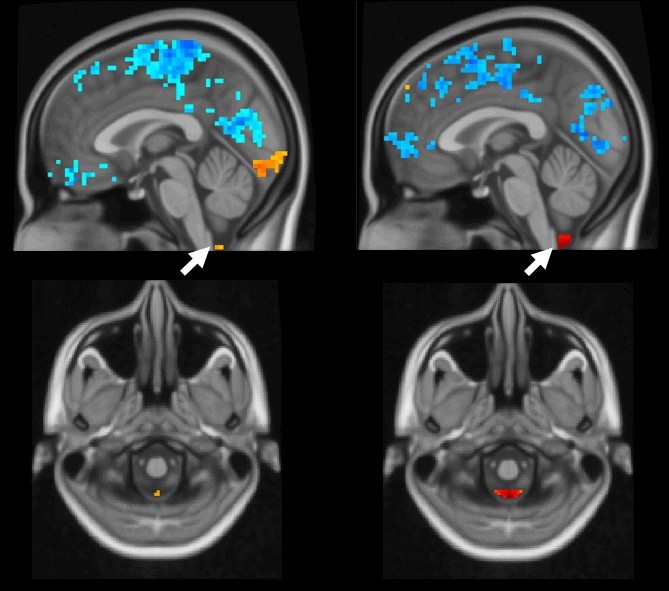
In human brains, as global brain activity (blue) occurs, the arrows point to cerebral spinal fluid moving (orange and red). In these functional MRI images, the left brain demonstrates a weaker coupling, indicating Alzheimer's disease. A healthy coupling appears in the right images. IMAGE: XIAO LIU/PENN STATE
Coupled brain activity, cerebrospinal fluid flow could indicate Alzheimer’s risk
Posted on June 2, 2021UNIVERSITY PARK, Pa. — Penn State researchers may have discovered a potential marker to clinically evaluate patients’ risk for Alzheimer’s disease through non-invasive imaging tests, according to a study published today (June 1) in PLOS Biology. The finding may have implications for diagnosis and treatment of the disease that results in significant cognitive decline, the researchers said.
Using functional magnetic resonance imaging (fMRI) data, the scientists found that brain activity is coupled to cerebrospinal fluid movement in humans. In fMRI, a person’s brain activity can be non-invasively measured by a blood oxygen level-dependent (BOLD) signal — blood moves to the active part of the brain, lighting up as it goes. In healthy individuals, once the BOLD signal flashes globally throughout the brain, cerebrospinal fluid flows from the base of the brain in rapid succession.
“The weaker the coupling of the global BOLD signal and cerebrospinal fluid flow, the higher the risk appears for Alzheimer’s disease,” said Xiao Liu, assistant professor in the Department of Biomedical Engineering and faculty co-hire in the Institute of Computational and Data Sciences, who led the study. “This could be a potential imaging marker for neurodegenerative diseases, as well as a non-invasive assessment approach for the recently discovered glymphatic system that helps clear toxins from the brain.”
First described in 2012, the glymphatic system acts as a waste management system, washing out the proteins and other buildup that can hinder brain activity. The critical component of the system is the cerebrospinal fluid flow, according to Liu, which his research suggests is activated by the global BOLD signal activation. In sleep, the glymphatic system can speed up its cleaning processes significantly.
“We saw the strongest coupling in subjects who were lightly sleeping based on other ongoing research in the lab,” Liu said, noting that they were not specifically studying the coupling in sleep but called the occurrence interesting, nonetheless. “We know sleep can promote the process of the glymphatic system and help clean out brain toxins. It looks like it could be through this coupled mechanism.”
To better understand the nuances of sleep and the coupled signaling, Liu collaborated with Orfeu M. Buxton, Elizabeth Fenton Susman Professor of Biobehavioral Health in the College of Health and Human Development, who directs the Sleep, Health, and Society Collaboratory at Penn State.
“The glymphatic system is highly related to sleep — we know that from intensive research conducted in animal studies,” said Buxton, who co-authored the study. “This new work expands the limited evidence base for evaluating the importance of this system in humans. This model, based on non-invasive imaging, has the potential to apply to anyone in any population to assess the activity of their glymphatic system.”
This research supports the emerging idea that a lack of sleep, a common issue for people with Alzheimer’s disease, could contribute to this reduced coupling, Liu and Buxton said. While it is not clear if sleep problems are a cause or a symptom of Alzheimer’s disease, the suggestion supports another recent international study that found people who sleep six hours or less were about 30% more likely to be diagnosed with dementia, another common symptom in people with Alzheimer’s disease, than those who sleep seven hours or more a night.
“Sleep is complicated, but this may be an avenue to begin understanding how sleep problems and Alzheimer’s disease influence each other,” Buxton said.
In addition to identifying and understanding how the coupled mechanism works, Liu said, the connection may also be a way to begin treating the disorder.
“Potentially, we could develop a therapy to increase the brain activity, which would increase the cerebrospinal fluid flow to more efficiently and regularly clear the toxins,” Liu said. “That’s purely theoretical at this point, but there is value in investigating new approaches.”
The team used human data collected by the Alzheimer’s Disease Neuroimaging Initiative (ADNI), a longitudinal, multi-site study launched in 2004 to investigate early detection and tracking of Alzheimer’s disease and facilitating data sharing between researchers. Currently, the initiative includes scientists from 59 research centers across the United States and Canada, who have collected varied data on almost 2,000 human subjects. Fewer than 150 subjects fit Liu’s study parameters of longitudinal fMRI information on the BOLD and cerebrospinal fluid movements.
“We’re continuing to investigate this link in other cognitive disorders, as well as in other types of data, such as genetics and other imaging approaches,” Liu said.
Co-authors include Feng Han, Jing Chen, Aaron Belkin-Rosen and Yameng Gu, all with the Department of Biomedical Engineering; and Liying Luo, assistant professor of sociology and criminology and in the Population Research Institute in the College of the Liberal Arts.
The National Institutes of Health and the White House Brain Research through Advancing Innovative Neurotechnologies (BRAIN) Initiative supported this work. The data collection for this project was funded by the ADNI, which is funded by the National Institutes of Health and the Department of Defense.
